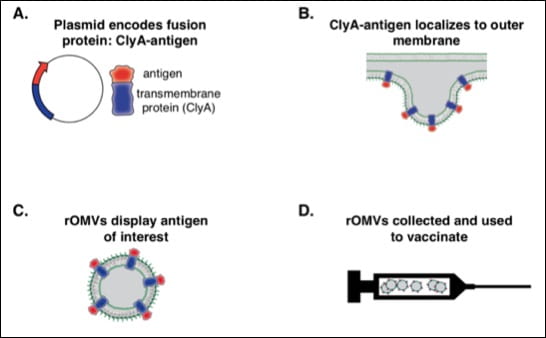
Development of vaccines for influenza is the largest arm of the research done in Putnam lab. Vaccines prevent disease by providing an acquired immunity to the body, which can then fight off particular infections. Most vaccines contain the antigen of interest from the bacteria or virus that is the target pathogen, and adjuvants that help elicit an enhanced immune response. The crux of out research hinges on the fact that bacteria produce small buds called outer membrane vesicles (OMVs, images below) that also produce an immune response. These OMVs can be used as adjuvants themselves, while also expressing recombinant antigen on their surface to target the pathogen of interest. Thus, they provide dual activity in one compact package, unlike current vaccines which contain a simple mixture of aluminum compound for adjuvant activity and antigen for pathogen activity. Being able to target both the adjuvant and antigen to the appropriate immune cells together provides a novel and promising way to develop new vaccines.
Transmission electron microscopy (TEM) images of OMVs.
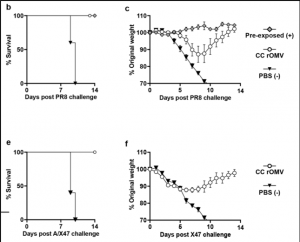
In our previous work, we have shown that M2e, the antigen associated with influenza, can be expressed on the surface of the OMVs using the transmembrane protein ClyA. This vaccine platform showed similar protection in mice that were pre-exposed to influenza before being challenged (image below), indicating that this could be a viable vaccine platform. In future, we are doing more work to conjugate various ligands to target different receptors of the immune system, including the various TLRs and NOD-like receptors, to obtain a targeted immune response for each individual infection in order to minimize side effects and produce a robust immune response.